Devices
This page outlines the considerations regarding devices as part of a successful Concentric digital consent deployment across an organisation.
What devices does Concentric work on?
Concentric works on any modern web-enabled device that meets our browser support policy. This includes full functionality across all screen sizes (i.e. mobiles, tablets, laptops and desktop computers).
Do we need to buy devices as part of introducing Concentric?
Most deploying organisations do not buy additional hardware purely as part of introducing Concentric. Initially, the deploying organisation should consider their existing hardware and consider whether it can be used for Concentric and current use cases.
Examples we see:
- Computer/Workstation on Wheels (WOWs) used for bedside consent conversations
- Tablets (iPads or otherwise) used for patient observations also being used for digital consent
In our experience, many clinicians prefer to use their mobile phones to use Concentric, knowing that it is always available and fast. No patient data is stored on the device, and access is secure.
There are some situations where we do see organisations more commonly purchasing additional devices:
- High volume areas such as endoscopy, where processes can often be successfully introduced around accessing and charging tablet devices without significant risk of device loss
- Certain patient populations where tablet devices are beneficial as a patient-held device, rather than using a computer screen at the end of the desk - for example in ophthalmology services.
Can we use signature pads with Concentric?
Some organisations have purchased, and get on well with, signature pads connected to desktop computers in outpatient departments and placed on WOWs for ease of signing on the ward. While these are not required - a signature can be added via QR handoff to a touchscreen device (either the patient or the clinician’s device) - a signature pad can be easier in some cases.
We formally support the Wacom STU-430 signature pad. Once paired, the option to “Sign using a signature pad” will be presented as the default option on the signing screen.
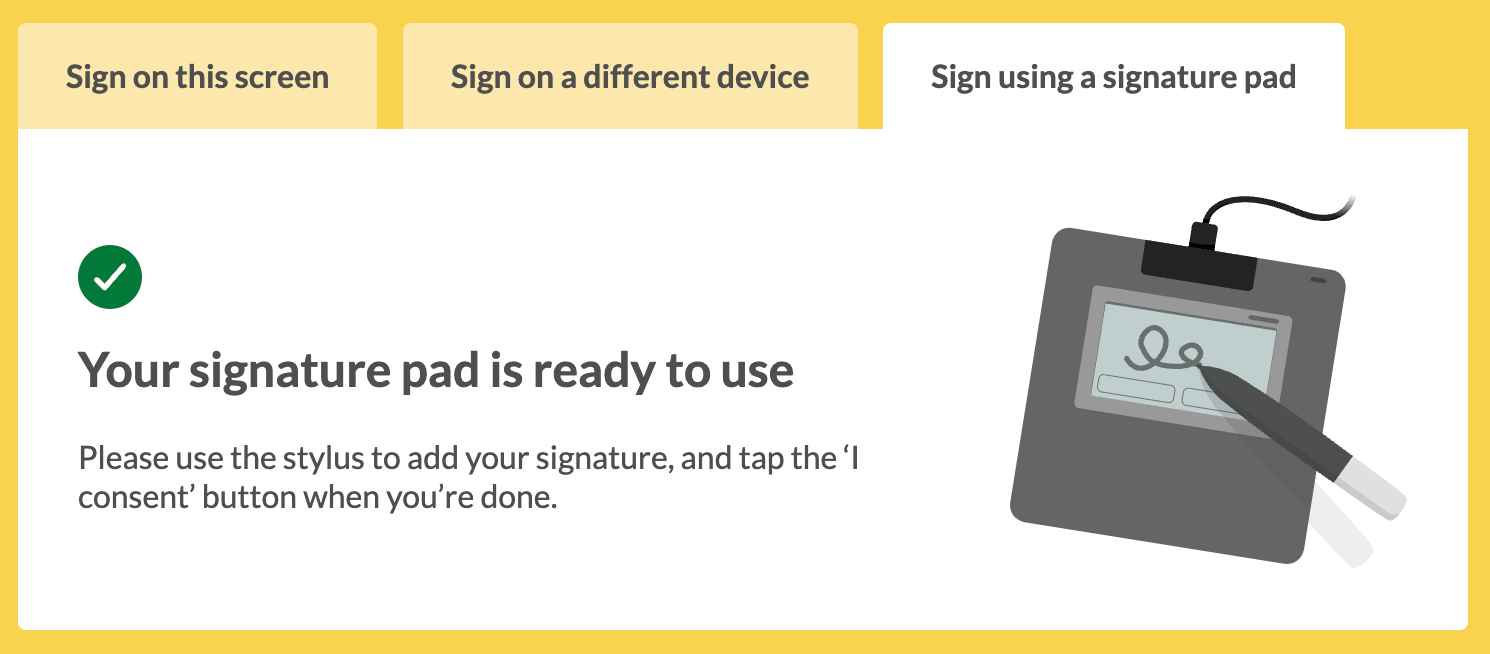
Read our setting up and using a signature pad page for full details of how to setup and use a Wacom STU-430 as part of your Concentric workflow.
Device related questions in our knowledge bank
Our knowledge bank answers common questions about Concentric from clinicians and deploying teams, and includes some device related queries: