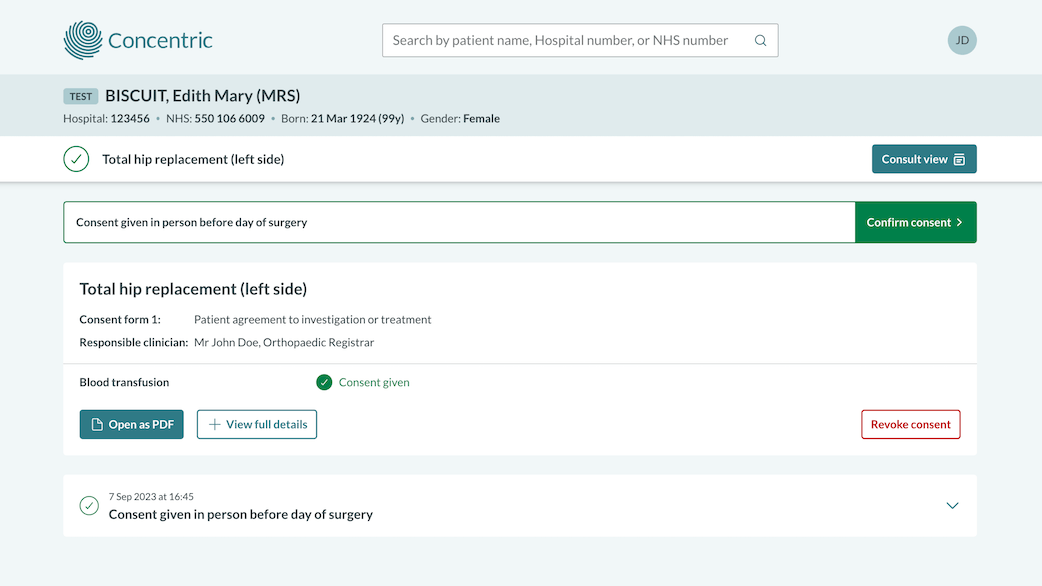
Why switch to digital consent?
Consent for treatment is a key element of care but the traditional paper-based consent process is no longer fit for purpose. It leads to medicolegal risk, system inefficiencies, and doesn't support shared decision making.

Reduce your medicolegal risk
An evolving medicolegal landscape means that the consent process - driven by paper-based consent forms often completed on the day of treatment - represents an increasing proportion of medico-legal risk.
Cases such as the Montgomery judgement have highlighted the need to improve consent practices and set in law standards around personalisation, documentation, time for consideration, and the necessity to provide information in plain language.
NHS Trusts have estimated that introducing digital consent reduces their 'failure to warn' (i.e. informed consent) risk by 50%.

Deliver on your shared decision making intentions
Improving shared decision making (SDM) quality has been shown to lead to better - often more conservative - decisions, more informed and engaged patients, and better outcomes.
Published evidence shows that introducing a digital consent process is one of the most effective mechanisms of driving shared decision making quality - an increase from 28% to 72% of patients reporting gold-standard SDM.

Fewer day-of-treatment delays
Paper-based consent processes are a major contributor to day-of-treatment delays. This is in part due to lost forms, and partly due to consent conversations occurring on the day of treatment, with both hampering elective recovery efforts.
Work by Imperial College Healthcare NHS Trust showed that day-of-surgery cancellations were reduced by 5-10%, and clinical time was released, once digital consent was embedded.

Save money and cut carbon
In many organisations, consent is one of the last remaining paper processes. This leads to significant costs relating to scanning or paper storage, and the piles of forms and patient information leaflets are contributing to a larger carbon footprint.
Health economics analysis demonstrates that, at most organisations, introducing digital consent is a cost-saving change. Further cost savings can be expected in the medium term with improved efficiency and reduction in medicolegal risk.
The carbon impact of the consent process is essentially removed thanks to net-zero cloud hosting.
To dive deeper into the evidence regarding digital consent, head to our digital consent research overview.
Why choose Concentric?
Considering digital consent options? Concentric leads the way.
“From paper to econsent. What's not to like?”